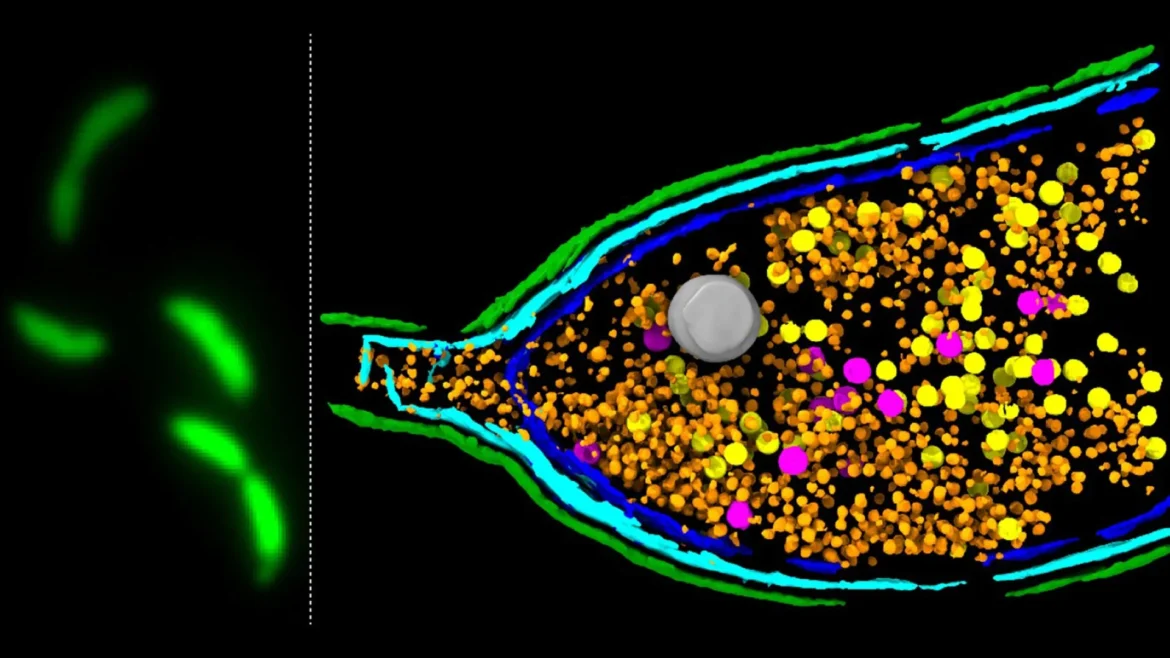
Scientists at the John Innes Centre have unveiled critical new insights into the clandestine mechanisms by which bacteria share genes, including those that fuel the escalating global health crisis of antimicrobial resistance (AMR). The groundbreaking research, published in the esteemed journal Nature Microbiology, illuminates the intricate workings of gene transfer agents (GTAs) – enigmatic particles that act as sophisticated couriers, disseminating genetic material between bacterial cells. This discovery provides a crucial piece of the puzzle in understanding how antibiotic resistance genes spread with alarming speed, posing a significant threat to modern medicine.
The Unseen Architects of Genetic Exchange
Gene transfer agents (GTAs) represent a fascinating adaptation within the bacterial world. Though they bear a striking resemblance to bacteriophages, the viruses that typically infect and exploit bacteria, GTAs are not inherently harmful invaders. Instead, they are believed to be derived from ancient viral lineages that bacteria have ingeniously co-opted and brought under their own metabolic control. This domestication has transformed potentially destructive entities into vital tools for bacterial evolution and adaptation.
These remarkable particles function as minuscule, highly specialized delivery vehicles. Their primary role is to encapsulate and transport fragments of DNA from one bacterial cell to others in its immediate vicinity. This fundamental process, known as horizontal gene transfer (HGT), allows bacteria to acquire and share beneficial traits with unprecedented efficiency. Among the most critical of these traits are genes that confer resistance to antibiotics, rendering life-saving drugs ineffective and complicating the treatment of bacterial infections worldwide. The implications of this rapid genetic dissemination are profound, as a single resistant bacterium can quickly seed a population with resistance genes, undermining years of antibiotic development.
The Crucial Step: Host Cell Lysis and GTA Release
A pivotal, yet until recently poorly understood, stage in the GTA lifecycle is host cell lysis. This is the dramatic process where the bacterial host cell ruptures, releasing the newly packaged GTA particles into the extracellular environment. The precise molecular machinery responsible for initiating and controlling this critical lytic event has remained a subject of intense scientific inquiry. Without an effective release mechanism, the gene transfer capabilities of GTAs would be severely curtailed, limiting their impact on bacterial populations. The ability of bacteria to orchestrate such a self-destructive event for the benefit of their genetic propagation highlights a remarkable evolutionary strategy.
Unmasking the LypABC Lysis Control System
Through a meticulous deep sequencing-based screening methodology, the research team at the John Innes Centre, in collaboration with esteemed colleagues from the University of York and the Rowland Institute at Harvard, successfully pinpointed the genetic components governing GTA activity. Their investigations focused on the well-characterized model bacterium Caulobacter crescentus, a common inhabitant of freshwater environments known for its complex cell cycle and genetic tractability.
The researchers identified a critical three-gene system, designated LypABC, responsible for producing specific bacterial proteins. The functional significance of this gene cluster became strikingly clear when it was manipulated. When the lypABC genes were experimentally deleted from the bacterial genome, the cells were rendered incapable of undergoing lysis, and consequently, no GTA particles could be released. Conversely, when the LypABC system was deliberately overactivated, a significantly higher proportion of bacterial cells underwent lysis, facilitating the widespread release of GTAs. These compelling results unequivocally demonstrate that the LypABC system acts as a central regulatory hub, orchestrating the controlled breakdown of the bacterial cell to permit GTA egress.
A Surprising Repurposing: From Immune Defense to Gene Transfer
Perhaps one of the most intriguing and unexpected revelations of this study is the striking molecular resemblance between the LypABC system and a known bacterial defense mechanism against phages – an anti-phage immune system. The protein components encoded by LypABC share significant structural and functional similarities with systems bacteria typically employ to defend themselves against viral invaders.
However, in the context of GTAs, this ancient defense machinery appears to have been ingeniously repurposed. Instead of solely serving a protective role, the LypABC system has been co-opted by the bacterium to facilitate its own genetic dissemination. This adaptation represents a profound example of evolutionary ingenuity, where existing biological pathways are modified and utilized for novel purposes. It underscores the remarkable flexibility and adaptability inherent in bacterial biology, allowing them to innovate and thrive in diverse and challenging environments.
The Imperative of Tight Regulation for Bacterial Survival
The research team also identified a crucial regulatory protein that plays a vital role in maintaining stringent control over GTA activity. This regulatory oversight is not merely a matter of efficiency; it is essential for the survival of the bacterial cell itself. The LypABC system, while instrumental for gene transfer, is also inherently toxic if improperly activated. Uncontrolled lysis can lead to the premature death of the bacterial cell, hindering its ability to replicate and contribute to the population. Therefore, the precise temporal and quantitative regulation of LypABC is paramount, ensuring that lysis occurs only when it is advantageous for gene transfer and at a controlled rate that does not compromise the overall viability of the bacterial community.
Broader Implications for Understanding Bacterial Evolution and AMR
This comprehensive study offers a deeper and more nuanced understanding of the dynamic processes by which genes move between bacterial cells. By shedding light on the molecular underpinnings of GTA-mediated gene transfer, the findings provide critical insights into the mechanisms driving the alarming spread of antibiotic resistance. The ability of bacteria to rapidly acquire and share resistance genes through HGT is a primary factor contributing to the emergence of multidrug-resistant pathogens, a phenomenon that threatens to revert healthcare to a pre-antibiotic era.
Dr. Emma Banks, the first author of the study and a Royal Commission for the Exhibition of 1851 Research Fellow at the John Innes Centre, emphasized the significance of these findings: "What’s particularly interesting is that LypABC looks like an immune system, yet bacteria are using it to release GTA particles. It suggests that immune systems can be repurposed to help bacteria share DNA with each other — a process that can contribute to the spread of antibiotic resistance." This observation highlights the complex interplay between defense mechanisms and genetic exchange within bacterial communities.
Future Directions and the Ongoing Fight Against Resistance
The current research has laid a robust foundation for future investigations. The immediate next step for the scientists is to delve deeper into the precise mechanisms that trigger the activation of the LypABC system. Understanding how this intricate control hub is switched on and how it meticulously governs the rupture of bacterial cells to release GTA particles will be crucial. Such knowledge could potentially lead to novel strategies for disrupting or inhibiting this vital gene transfer process.
The work conducted by the John Innes Centre, in partnership with the University of York and the Rowland Institute at Harvard, represents a significant stride forward in deciphering the evolutionary strategies of bacteria. By unraveling the secrets of gene transfer agents and their sophisticated control systems, researchers are gaining invaluable knowledge that could ultimately inform the development of new interventions to combat the ever-growing threat of antimicrobial resistance. The fight against AMR is a complex, multifaceted challenge, and understanding the fundamental biological processes that facilitate its spread is a critical component of any effective global strategy.
The implications of this research extend beyond the immediate concern of AMR. The discovery of repurposed immune systems for gene transfer sheds light on the remarkable plasticity of bacterial biology and the diverse evolutionary pathways they can exploit. This fundamental understanding of bacterial adaptation and genetic exchange is essential for fields ranging from microbial ecology to synthetic biology. As antibiotic resistance continues to loom large over public health, research like this offers not only crucial insights but also potential avenues for innovative solutions.